 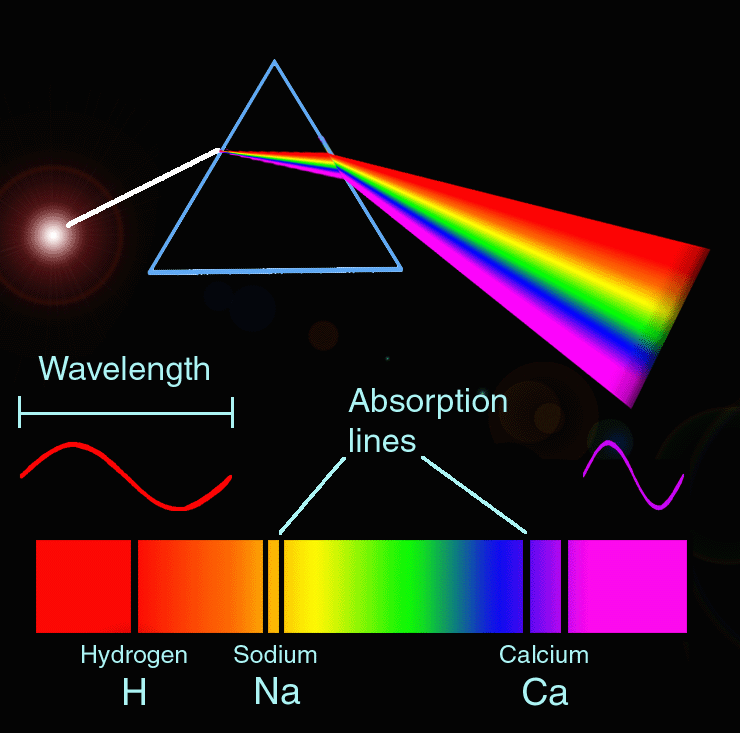
When a metal atom is changed into gas and light is passed from the sources, the ground state of the atom gets excited by absorbing the radiation of a particular wavelength. The process by which gaseous metal atoms are produced into the flame can be illustrated as: If the light of resonance wavelength is passed through the flame containing the atoms which are analyte, the part of the light will be absorbed and the extent of absorption will be directly proportional to the number of ground state atoms present in the flame. A large number of the gaseous metal atom remains in the ground state, and are capable of absorbing radiant energy of their specific wavelength. 
If a solution containing metal salt (M +X –) is aspirated to the flame, a vapor that contains atoms of metal may be formed. Principle of atomic absorption spectroscopy The modern form of AAS was developed by Australian Chemist, Sir Alan Walsh in the 1950s. Because the atomic absorption method is largely free of interference and the set of electronic energy levels is specific to that element, it is a highly good analytical technique with great sensitivity. It is used to determine the concentration of metals present in a sample to be analyzed.ĪAS can be used to quantify more than 70 different elements either in solution or solid form and possesses wider applications in clinical analysis, food analysis, the pharmaceutical industry, the mining sector, and so on. Difference between flame photometry and atomic absorption spectroscopyĪtomic absorption spectroscopy (AAS) is an absorption spectroscopic method that uses the absorption of light by free atoms in a gaseous state to determine the quantitative composition of chemical components.Disadvantages of atomic absorption spectroscopy.Advantages of atomic absorption spectroscopy.Application of atomic absorption spectroscopy.Interferences in atomic absorption spectroscopy.Atomic absorption spectroscopy instrumentation.Principle of atomic absorption spectroscopy. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
